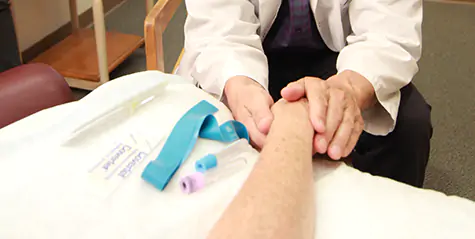
At York Hospital, we believe patients should have access to clinical trials close to home, so we offer both oncology and hematology. Our hope is to improve the lives of cancer patients by enrolling them in clinical trials that would help to solve important, challenging, and neglected problems in cancer research and patient care.
Maine Cancer Genomics Initiative
We have partnered with the Maine Cancer Genomics Initiative (MCGI), a special alliance of cancer experts, clinicians and researchers from The Jackson Laboratory, to offer genomic testing for patients and also to participate in studies that help highlight the best treatments for colon cancer, multiple myeloma and breast cancer.
Our team of providers, clinical trials nurse and clinical research assistant all work diligently to enroll qualified patients in active trials. We are proud of our involvement in, and support of, the ongoing need to enroll patients in both phase 2 and phase 3 trials.
Learn more about participating in clinical trials closer to home by contacting our office at (207) 351-3777.
What is a clinical trial?
A clinical trial is a research study designed to answer a question which is relevant to current patient care. Clinical trials are the major way advancements in medical care occur, and current cancer care is based on results from previous clinical trials. In order to conduct clinical trials, it is often necessary to perform extensive research in a laboratory to demonstrate that a particular drug or treatment is worthy of investigation.
Why participate in a clinical trial?
There are many benefits to clinical trial participation, both personal and societal. Some clinical trials are testing a new drug which has not yet been approved for cancer care by the FDA. Other clinical trials are designed to test new combinations of drugs or an approved drug in a cancer type in which it has not yet been studied.
How a clinical trial works
Clinical trials use written protocols which define the purpose of the trial, as well as the specifics of where and how it will be conducted. Clinical trials are extensively regulated for both ethical and medical purposes; each institution that offers clinical trials is required to maintain a board of representatives charged with the protection of human subjects. This board is known as an Institutional Review Board (IRB) and comprises individuals whose primary interest is the safe conduct of the trial at their institution.
Learn more about participating in clinical trials closer to home. Please contact our office at (207) 351-3777.
Clinical trials close to home
Our team works diligently to enroll qualified patients in active trials.